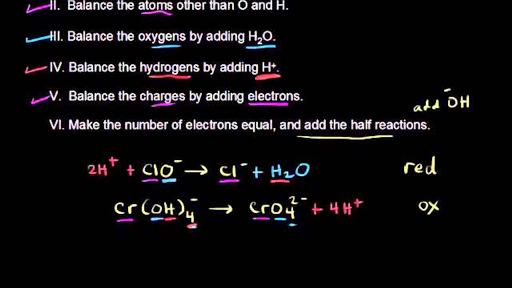MnO^ - 4 + Fe^2 + → Mn^2 + + Fe^3 + + H2O . (acidic medium)Balanced reaction is: MnO^ - 4 + aFe^2 + + bH^ + → Mn^2 + + cFe^3 + + dH2O .a + b + c + d is:
How to Balance Redox Equations in Acidic Solution - SchoolTube - Safe video sharing and management for K12
How to Balance Redox Equations in Basic Solution - SchoolTube - Safe video sharing and management for K12






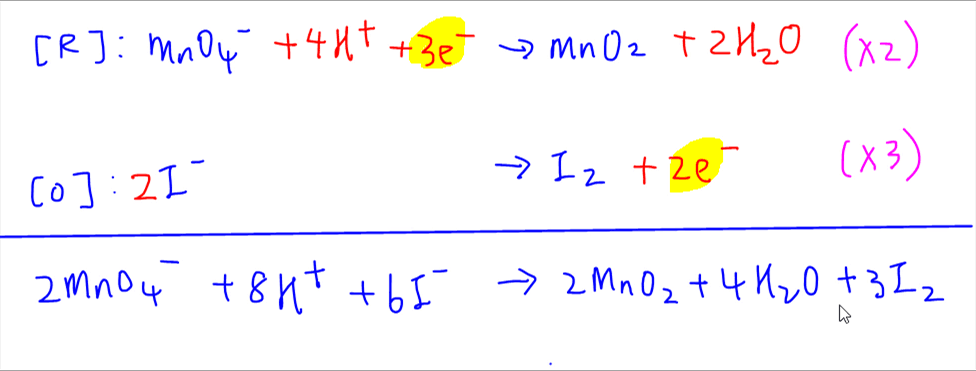

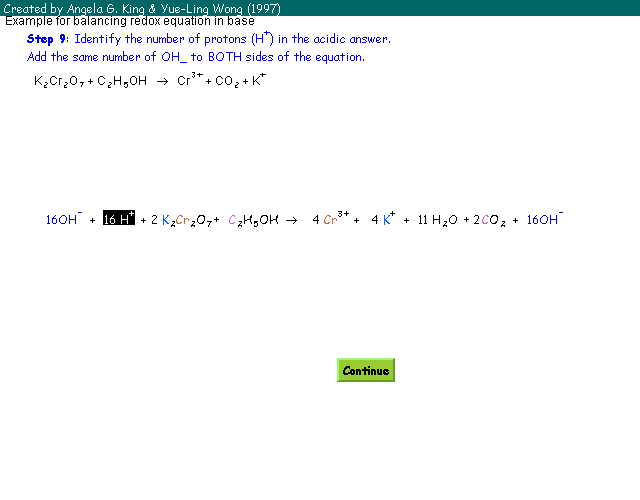
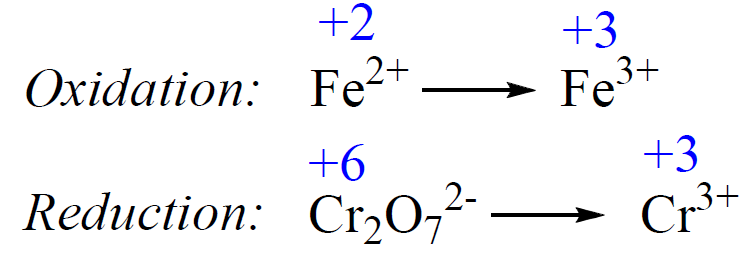






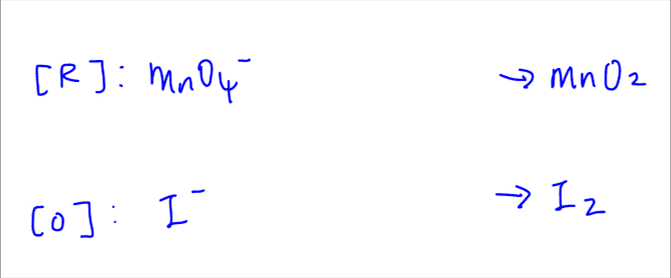

:max_bytes(150000):strip_icc()/redoxhalfreactions-56a12b323df78cf772680e96.jpg)